Battery Materials
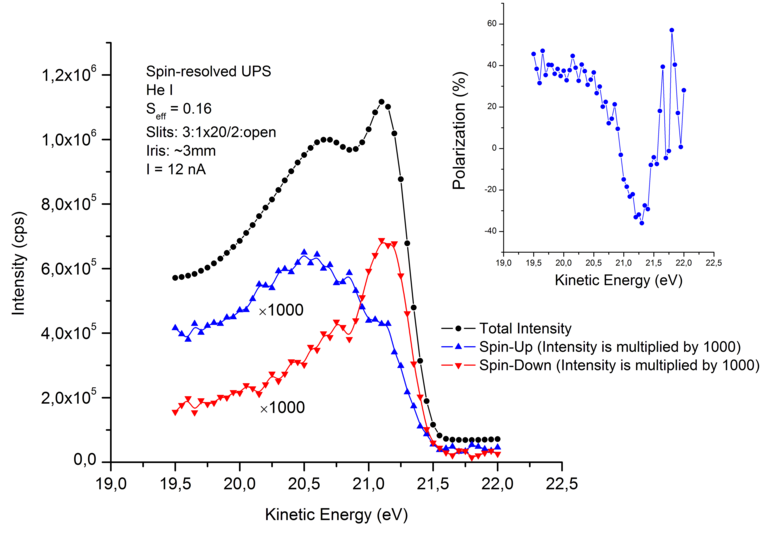
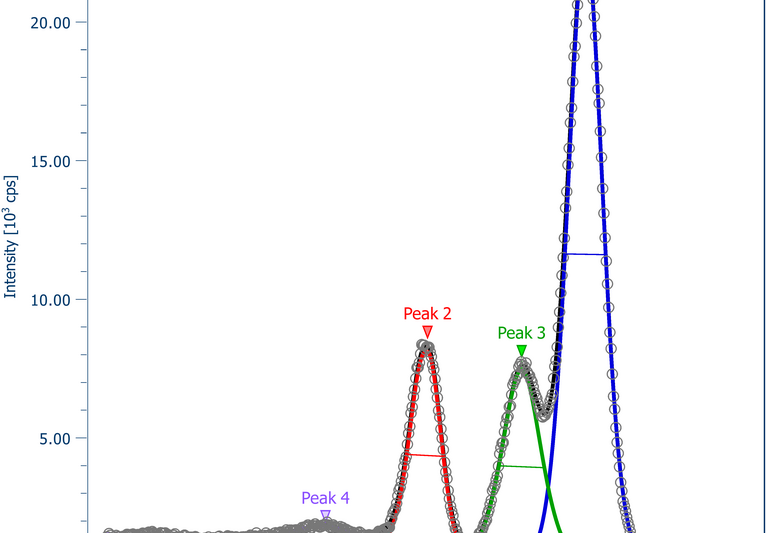
A secondary lithium ion battery consists of an anode, a cathode and an electrolyte (liquid or solid) separating both. While during charging and discharging cycles the electrons are shifted via the outer circuit from anode to cathode and vice versa, the electrolyte allows for the transfer of the ions into the cathode (discharging) or anode (charging). The potential difference between cathode and anode reflects the battery voltage. During this process films on both electrodes are formed at the interface between electrolyte and electrode. This can be beneficial for the stability or accelerate the degradation of the battery. The chemistry and the potentials can be characterized pretty well with (NAP)-XPS and HAXPES.
Your web browser is deprecated
This could effect the presentation and some functions of our website.